HIV Injection Costs $28,000 Annually, Sparking Debate on Accessibility
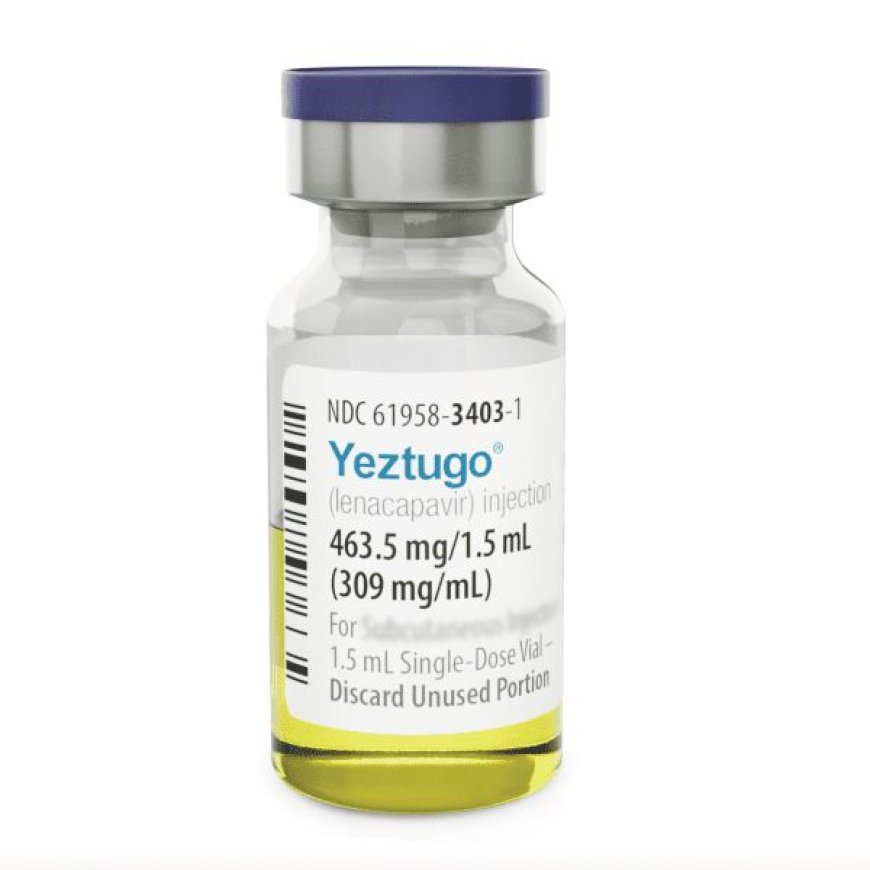
The US Food and Drug Administration has granted approval for Yeztugo, a groundbreaking twice-yearly injectable medication for HIV prevention, marking a significant milestone in the fight against the disease. Developed by Gilead Sciences, Yeztugo has shown remarkable efficacy in clinical trials, boasting a 99.9% success rate in preventing HIV transmission.
How Yeztugo Works
This innovative treatment belongs to a new class of HIV drugs called capsid inhibitors, which interfere with the virus's protective shell, preventing replication. Administered via subcutaneous injection every six months, Yeztugo offers a convenient and discreet prevention strategy for high-risk populations.
Clinical Trial Results
The FDA's approval was based on data from two large Phase 3 trials, PURPOSE 1 and PURPOSE 2, which demonstrated Yeztugo's exceptional effectiveness in diverse groups. In the trials:
- Zero HIV infections occurred among cisgender women receiving lenacapavir in Africa.
- Only two infections were recorded among men and gender-diverse individuals, resulting in a 99.9% prevention rate.
Pricing and Accessibility Concerns
Despite its potential, Yeztugo's high price tag has sparked controversy. With an estimated annual list price of $28,218 in the US, many worry that accessibility will be limited to the wealthy. However, experts suggest that generic versions could be produced at a significantly lower cost, around $25-$40 per patient annually, if manufactured at scale.
Global Impact and Future Outlook
Gilead has partnered with six manufacturers to produce cheaper versions of Yeztugo for 120 low-income countries, signaling a push toward global access. Nevertheless, logistical challenges, infrastructure readiness, and sustained funding will be crucial in determining the treatment's real-world success. As Dr. Monica Shah, a leading infectious disease specialist, notes, "The approval of lenacapavir marks a significant leap forward in the fight against HIV, but its real impact will only be felt if it's accessible to the people who need it most".
Source - Lead News Online















































)










